Troubleshoot your Westerns using the reference guide below, download the Protein Blotting Handbook for principles and protocols, or for. To start troubleshooting your western blotting problems, choose the type of problem you are experiencing. As your Western blotting partner, our technical support team is ready to help you analyze protein expression and protein-protein interactions with sensitivity and reproducibility. Comprehensive solutions and suggestions are provided to help solve your particular western blotting challenges. Always include protease inhibitors (and phosphatase inhibitors for the detection. Use fresh, sonicated, and clarified tissue extracts. Tissue extracts tend to produce more non-specific bands and degradation products. In this section, you can find solutions to issues related to protein band appearance. Use freshly prepared sample kept on ice up until the addition of sample buffer and immediate heating to 95☌ for 510 minutes. The Western Blot Doctor is a self-help guide developed by Bio-Rad researchers that enables you to identify and troubleshoot western blotting problems. The Western Blot Doctor is a self-help guide that enables you to troubleshoot your western blotting problems. The final concentration will be 5 v/v glacial acetic acid and 0.1 w/v Ponceau S. Tips, Techniques, and Technologies from the Western Blotting Experts at Bio‑Rad Laboratories: Get Your Free Guide To make a 100 mL solution from the powder, weigh 100 mg of Ponceau S powder and make it up to 95 mL with distilled water before adding 5 mL of glacial acetic acid. Comprehensive solutions and suggestions are provided to help solve your particular western blotting challenges.with the Western Blot Doctor™ Make sure that the methanol concentration in the transfer buffer is not more than 10–20% and that high-quality, analytical grade methanol is used.The Western Blot Doctor is a self-help guide developed by Bio-Rad researchers that enables you to identify and troubleshoot western blotting problems. It may cause a reduction in pore size, precipitation of some proteins, and some basic proteins to become positively charged or neutral. 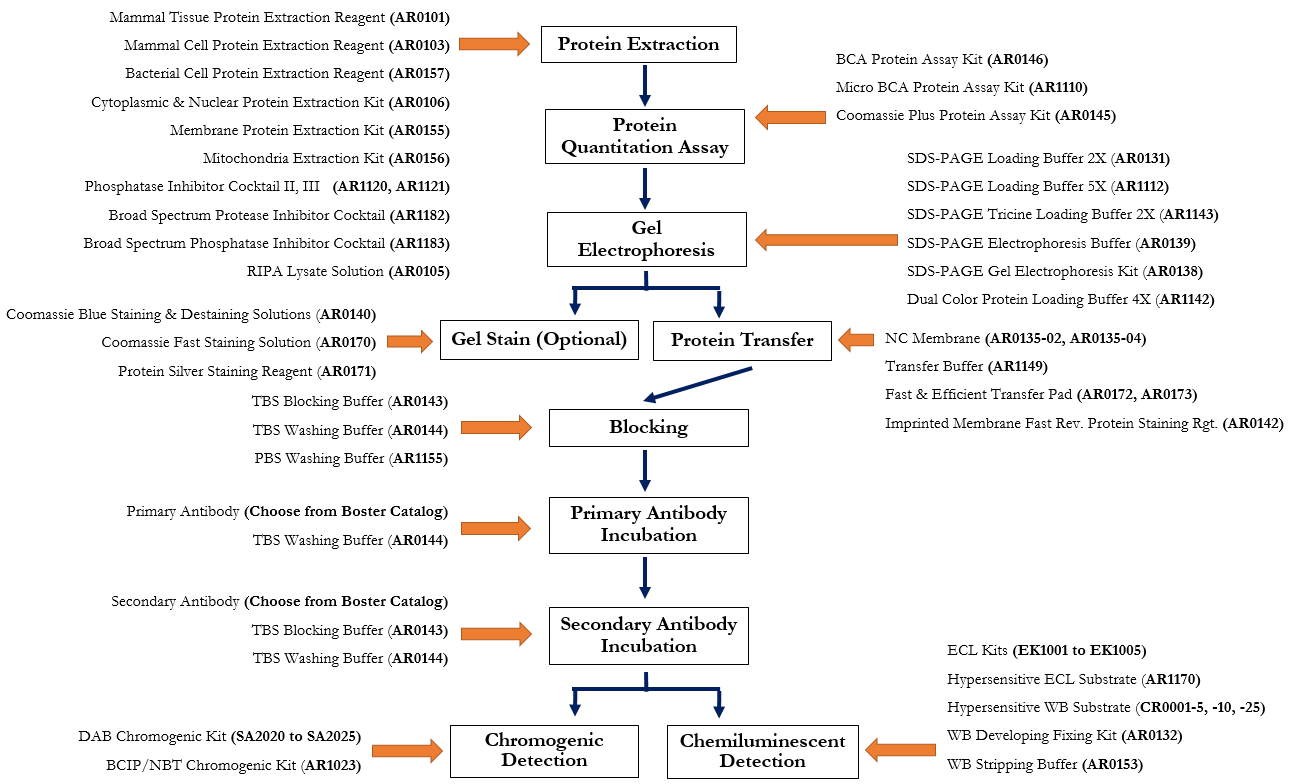
Methanol removes the SDS from SDS-protein complexes and improves the binding of protein to the membrane, but has some negative effects on the gel itself, leading to a decrease in transfer efficiency. 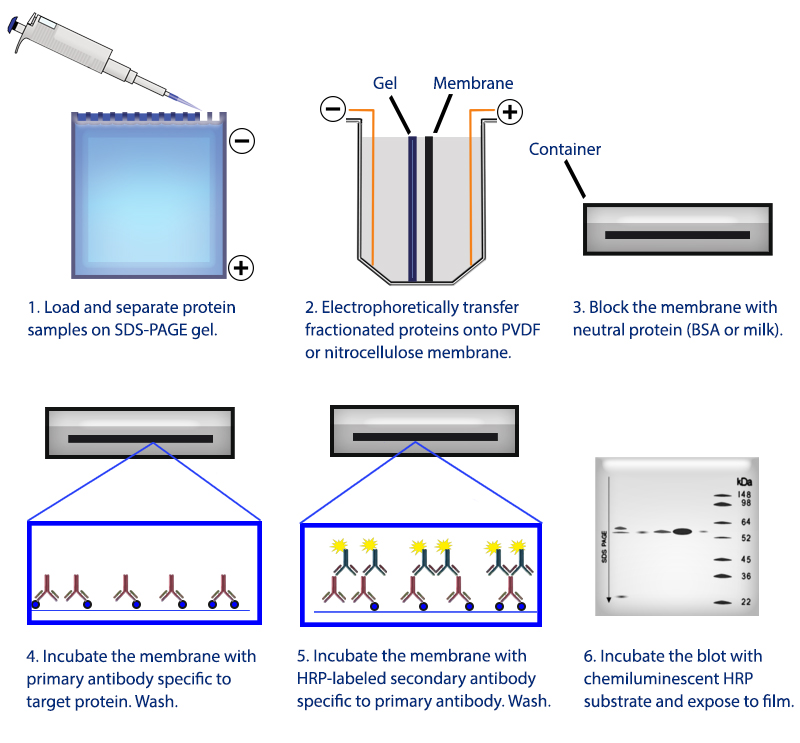
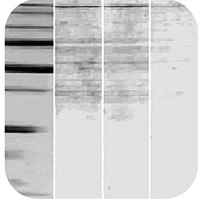
Want a better way Try our Thermo Scientific Restore PLUS Western Blot Stripping Buffer for efficient stripping in just 10 minutes at room temperature. Observation: Patchy uneven spots all over the blot Contaminated equipment, Make sure that the electrophoresis unit is properly washed. Use homemade stripping buffer and heat the blot at 37☌ for up to 30 minutes. Request a free Western blot tips, tricks and troubleshooting guide. We recommend pre-equilibrating the gel in 2x Transfer buffer (without methanol) containing 0.02–0.04% SDS for 10 minutes before assembling the sandwich and then transferring using 1x transfer buffer containing 10% methanol and 0.01%SDS. Always wet the blot with wash buffer for 510 minutes before adding stripping buffer. In this western blot troubleshooting section, we will help you visually identify specific and common problems on your western blots, such as high background, weak or no signal, multiple bands, uneven staining and suggest what may be causing them and some solutions to remedy them. For proteins that are difficult to elute from the gel such as large molecular weight proteins, a small amount of SDS may be added to the transfer buffer to improve transfer. This inhibition is higher for nitrocellulose than for PVDF.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
